Derma-Stent™
Derma-Stent™ Abscess Drain
A minimally invasive cutaneous abscess drain
For increasingly common abscesses, as well as cysts, seroma, hematoma, and more
- Comfortable
- Easy to Use
- Versatile Design
- Save Space
- Made in the USA
Using the Derma-Stent™
Click Here to see the Derma-Stent™ abscess drain in action.
Mar-Med’s Derma-Stent™ Abscess Drain:
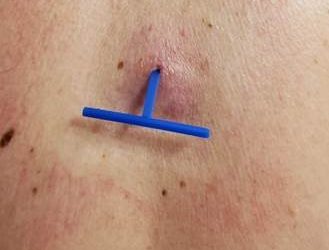
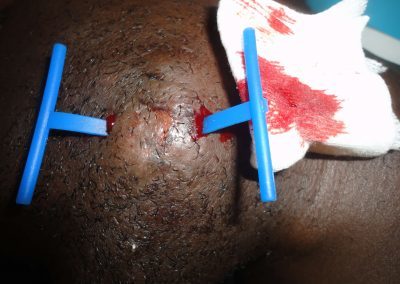
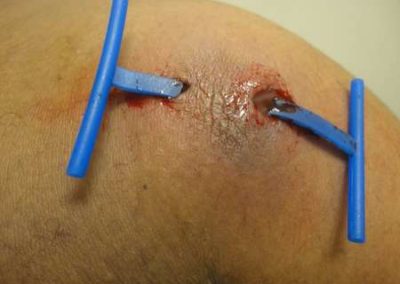
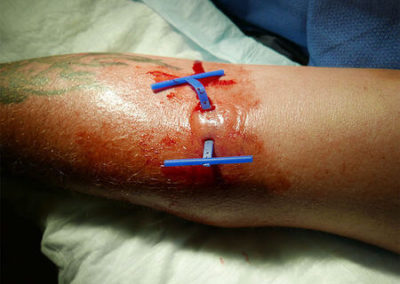
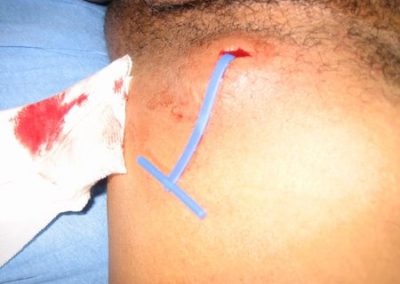
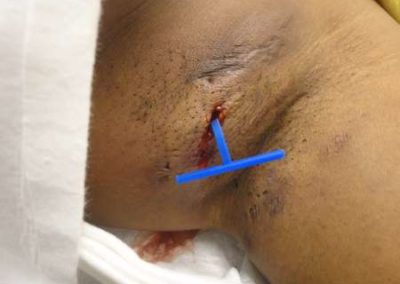
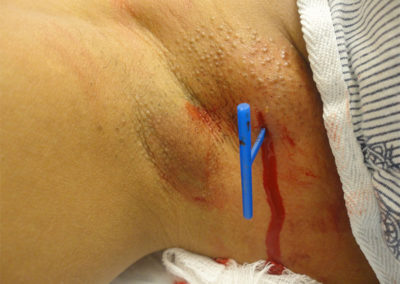
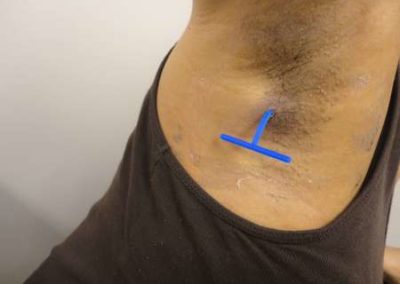
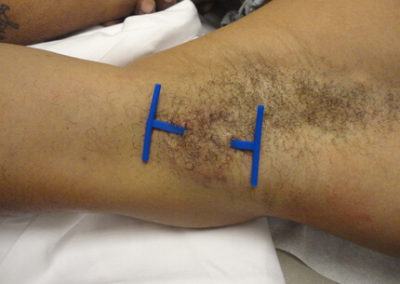
Derma-Stent is a new, minimally invasive cutaneous abscess drain preferred over packing gauze for packing an abscess by clinicians and patients alike in a recent trial. Clinicians are moving away from packing an abscess with gauze for incision and drainage because it’s painful, can cause toxic shock and often requires multiple visits for evaluation and management. Derma-Stent provides the ease and comfort of the “loop drain” method, but the unique design can be customized for use in 1 or 2 incisions, without tying clumsy knots or sutures. The Loop Drain method is becoming increasingly more common among urgent cares, emergency rooms and in pediatric settings due to the comfortability and ease of application to all kinds of patients.
Derma-Stent™ Clinical Trial Completed!
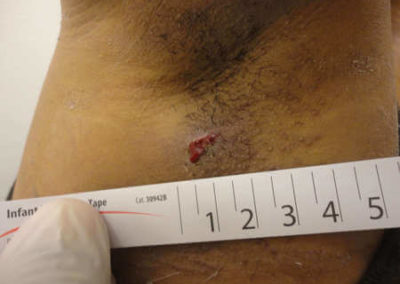
A randomized, controlled clinical trial (ClinicalTrials.gov Identifier: NCT03171714) comparing standard abscess packing with packing gauze to using Derma-Stent for abscess treatment was completed at Wayne State University under Aaron Brody, MD, MPH. The Journal of Emergency Medicine has published Dr. Brody’s description of using Derma-Stent on the trial’s first enrollee, which you can read about the here! And Open Access Emergency Medicine has published the full trial, which you can read about here! Lastly, view all of the images from the trial by scrolling through the gallery at the bottom of this page.
Instructions
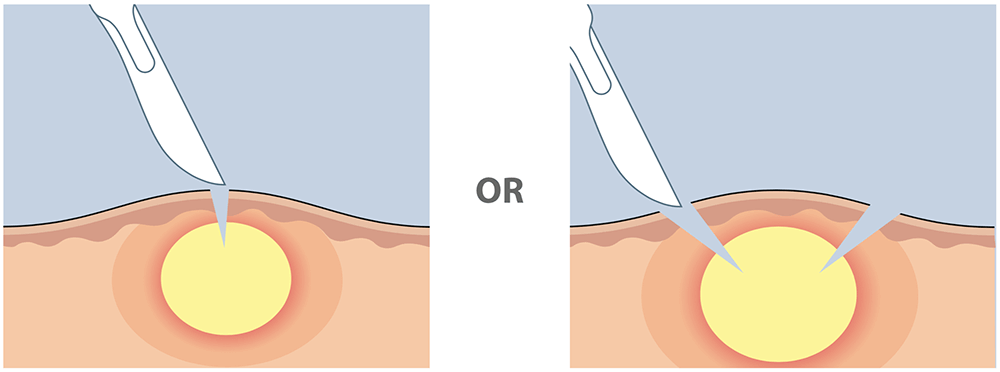
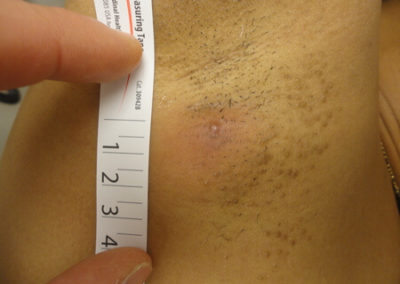
1) Drain the site using 1 or 2 incisions.
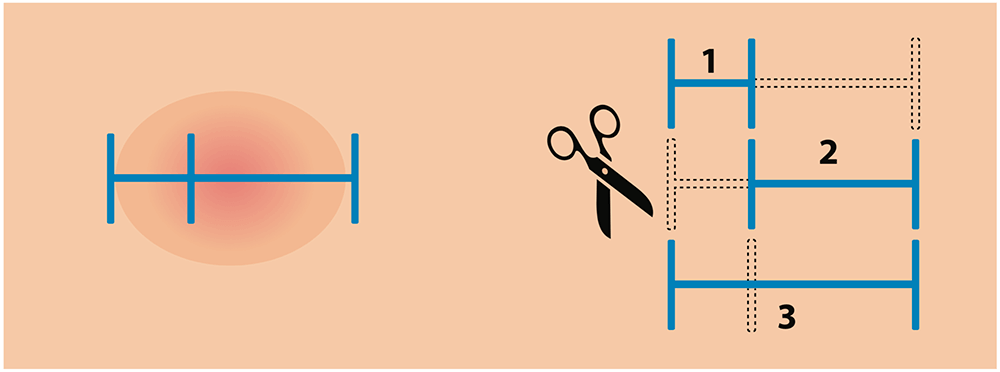
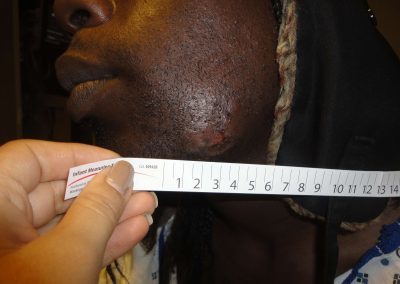
2) Measure the site along the Derma-Stent™. Cut to appropriate size.
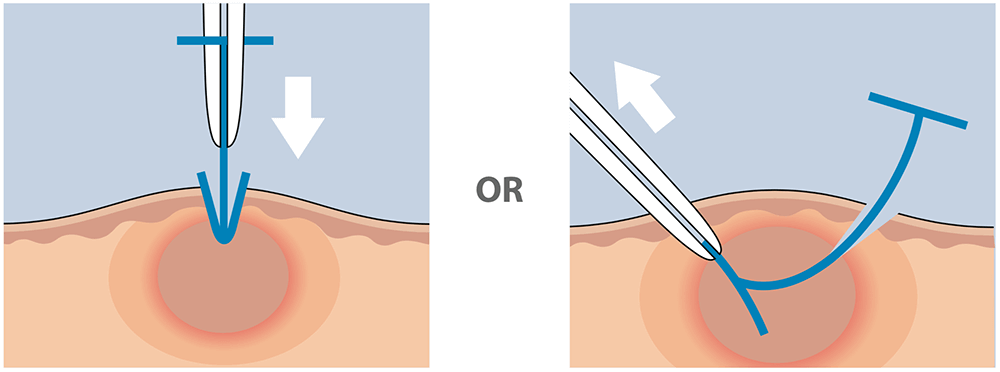
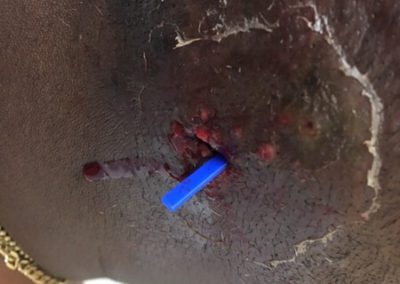
3) Insert Derma-Stent™ into cavity.
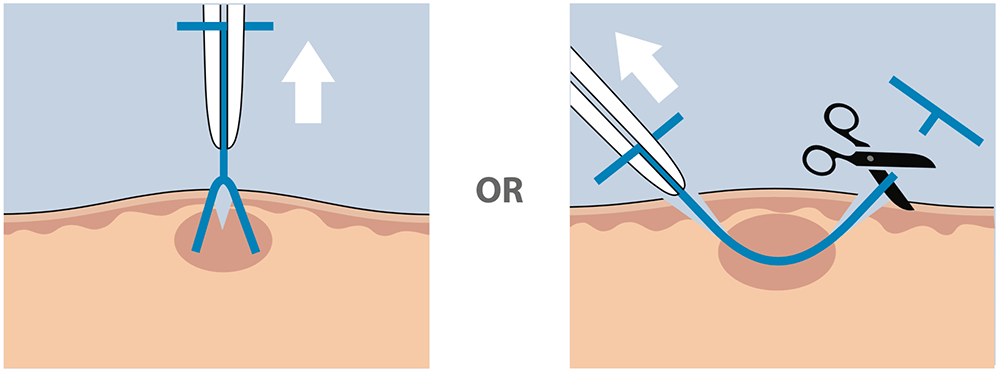
4) Remove Derma-Stent™ in accordance with clinician instructions.
Product Codes
DMT – cutaneous abscess drain.
Derma-Stent abscess drains come individually packaged and are latex free. Mar-Med minimum order is 1 box of 20 units.
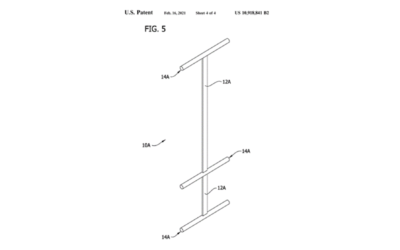
(Covered by US Patent No. 10,918,841 B2, US Patent No. D800902 and EUROPEAN Patent No. 003381771-0001.)
Derma-Stent™ Added Value
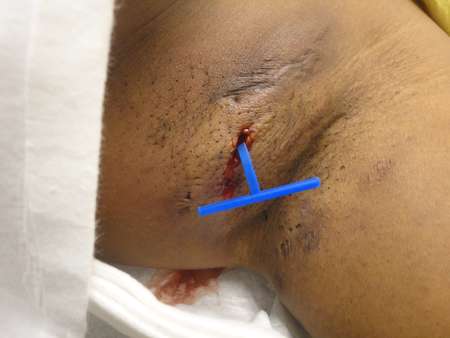
An estimated 4.5 million incision & drainage (I&D) procedures are performed on abscess cases in the USA each year in outpatient and acute care. For years abscess packing has been thought to keep abscess walls separated and allow drainage by preventing premature closure using packing gauze. But it is extremely painful, especially for pediatric patients, and often requires follow up and still has a relatively high failure rate. A new less invasive method, called the “Loop Drain Method”, is performed by tying the loop drain through two small incisions and meta-analysis of literature already suggests this method provides lower failure rates (4.10% vs 9.43%), potential for less pain, decreased scarring, and lower healthcare utilization costs. A similar technique involving a Jacobi ring has been demonstrated to compare well against the word catheter used on bartholin cysts. Derma-Stent is based off the Loop Drain Method and is easier to apply to various types of abscesses and cysts and proven to be less painful and scarring for pediatric patients.
Our Solution
Mar-Med’s Derma-Stent abscess drain was designed with the benefits of the minimally invasive method in mind, but simpler. A Clinical Trial has recently demonstrated the comfort and efficacy of our abscess drain. Whether for draining abscesses or cysts, in an adult or a pediatric patient, Derma-Stent should be considered to replace packing gauze for abscess packing, loop drains and word catheters, while saving cost and improving care.
Derma-Stent Abscess Drain Related Literature:
Randomized Controlled Trial of a Novel Silicone Device for the Packing of Cutaneous Abscesses in the Emergency Department: A Pilot Study, Brody A, Gallien J, Reed B, Hennessy J, Twiner MJ, Marogil J
- Summary: the silicone device was more likely to remain in place at day 3 follow-up and was equally effective to gauze packing in abscess reduction while also improving patient-reported pain scores.
A Novel Silicon Device for the Packing of Cutaneous Abscesses; Aaron M. Brody, MD, MPH, et al.
- Summary: In this single case, the provider found the device easy to use and noted that it did not extend the time of the procedure. The device was well tolerated by the patient and the patient was able to remove the device independently
A Randomized Controlled Trial of Novel Loop Drainage Technique Versus Standard Incision and Drainage in the Treatment of Skin Abscesses; Jay Ladde, Sara Baker, Natali Lilburn, Michelle Wan, Linda Papa
- Summary: The LOOP and packing techniques had similar failure rates for treatment of subcutaneous abscesses in adults, but the LOOP technique had significantly fewer failures in children. Overall, pain and patient satisfaction were significantly better in patients treated using the LOOP technique.
Loop Drainage Is Noninferior to Traditional Incision and Drainage of Cutaneous Abscesses in the Emergency Department, Elissa M. Schechter‐Perkins MD, MPH Kristin H. Dwyer MD, MPH Anish Amin MD Matthew D. Tyler MD James Liu MS Kerrie P. Nelson PhD Patricia M. Mitchell RN.
- Summary: The study provides evidence that Loop Drain Method is noninferior to I&D in achieving complete abscess resolution at 14 days and is associated with fewer return emergency room visits and fewer complications. This makes it an attractive alternative treatment option for abscesses.
Comparison of the loop technique with incision and drainage for soft tissue abscesses: A systematic review and meta-analysis; Gottlieb M, et al.
- Summary: A total of 1,374 studies were identified with eight studies (n = 910 patients) selected for inclusion. Overall, CID failed in 69 of 487 patients (14.17%), while LDT failed in 35 of 423 patients (8.27%). There was an OR of 2.02 (95% CI = 1.29 to 3.18) in favor of higher failures in the CID group.
Loop and drain technique for subcutaneous abscess: a safe minimally invasive procedure in an adult population; Gaszynski R, et al. ANZ Journal of Surgery. 2018 Jan;88(1-2):87-90 EPUB Sept 2016
- Summary: Loop Drain Technique…saves operating theatre time with 30% of Loop Drain Technique treated in the Emergency Department and ensures excellent follow-up compliance as patients return for VessiLoop removal. Healthcare burden is greatly reduced using Loop Drain Technique; most patients require only one review at 10-14 days.
Treatment of subcutaneous abscesses in children with incision and loop drainage: A simplified method of care; Aprahamian CJ, et al, Journal of Pediatric Surgery. Sep;52(9):1438-1441.
- Summary: The findings eliminate the need for repetitive wound packing and simplify postoperative wound care. Loop Drain Method offers shorter time to discharge, lower recurrence rates, and minimal scarring. Additionally, there is expected cost reduction.
Jacobi ring catheter treatment of Bartholin’s abscesses; Gennis P, et al. American Journal of Emergency Medicine. (May 2005) Volume 23, Issue 3, Pages 414–415.
- Summary: “Ease of insertion was similar for the 2 devices. Patient satisfaction was higher in patients treated with the ring catheter.”
Testimonials
“The patients report no significant discomfort from the placement or removal. Some say they didn’t even realize it’s there. Much better than packing and re-packing wounds!”
“Wonderful product, less pain, easy to use.”
Dr. Gaddis was a key influencer in the creation of our Derma-Stent™ product.
Commentary on the Derma-Stent on ERcast by Dr. Rob Orman
Derma-Stent Abscess Drain Blog Posts
Great Derma-Stent Review by Shannon Neer!
Sometimes we receive great device feedback that simply has to be shared. Shannon Neer contacted Mar-Med a few months ago introducing herself as a clinician doing a high volume of abscess cases in Yakima, WA. She is a fan of the loop drain method and requested...
Derma-Stent Clinical Trial
About 5 years ago, Mar-Med developed the Derma-Stent and began a clinical trial at Wayne State University. We sought to compare the Derma-Stent to traditional incision and drainage with packing gauze. The results from that clinical trial were recently published by...
Derma-Stent Patent Granted to Mar-Med!
Mar-Med is excited to announce the issuance of utility patent #10,918,841 for the Derma-Stent device by the United States Patent and Trademark Office! The Derma-Stent is a unique cutaneous abscess treatment device, which I designed as an alternative to the...